We, as clinical research professionals, strive to create and provide reliable healthcare information through our business activities below.
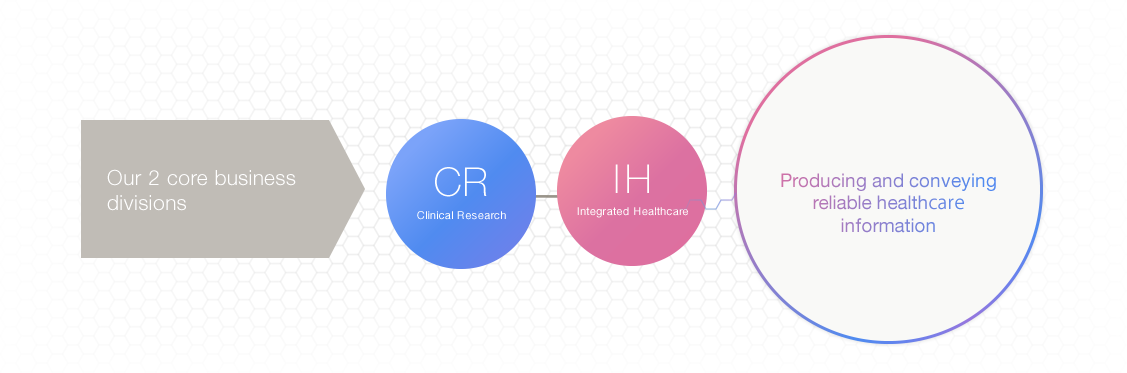
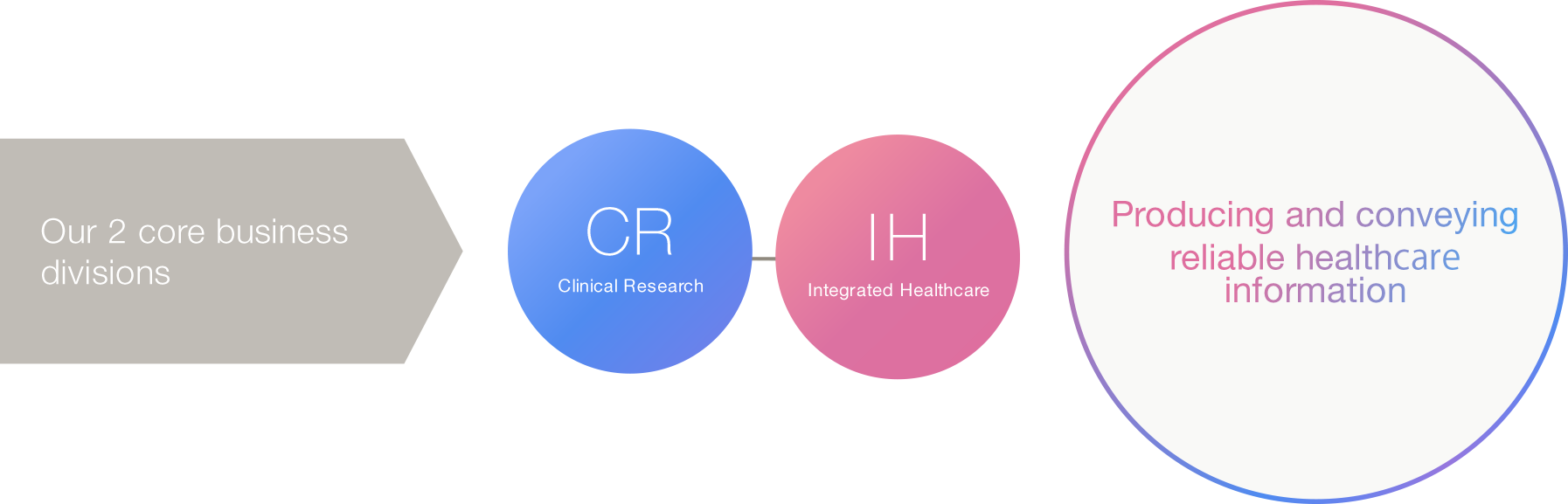
We, as clinical research professionals, strive to create and provide reliable healthcare information through our business activities below.

